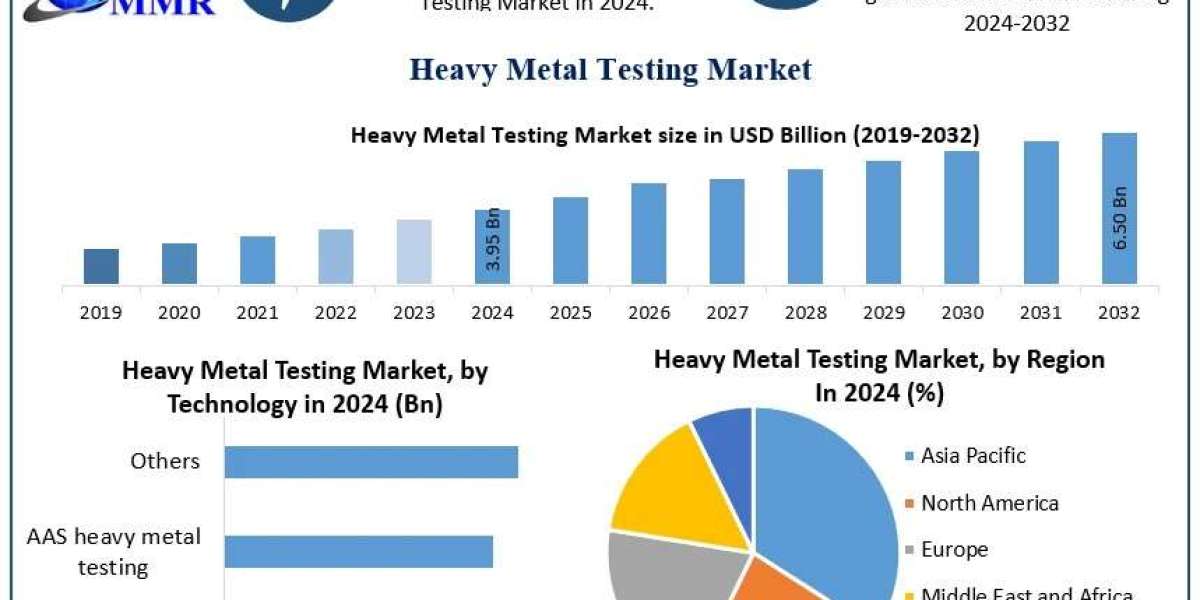
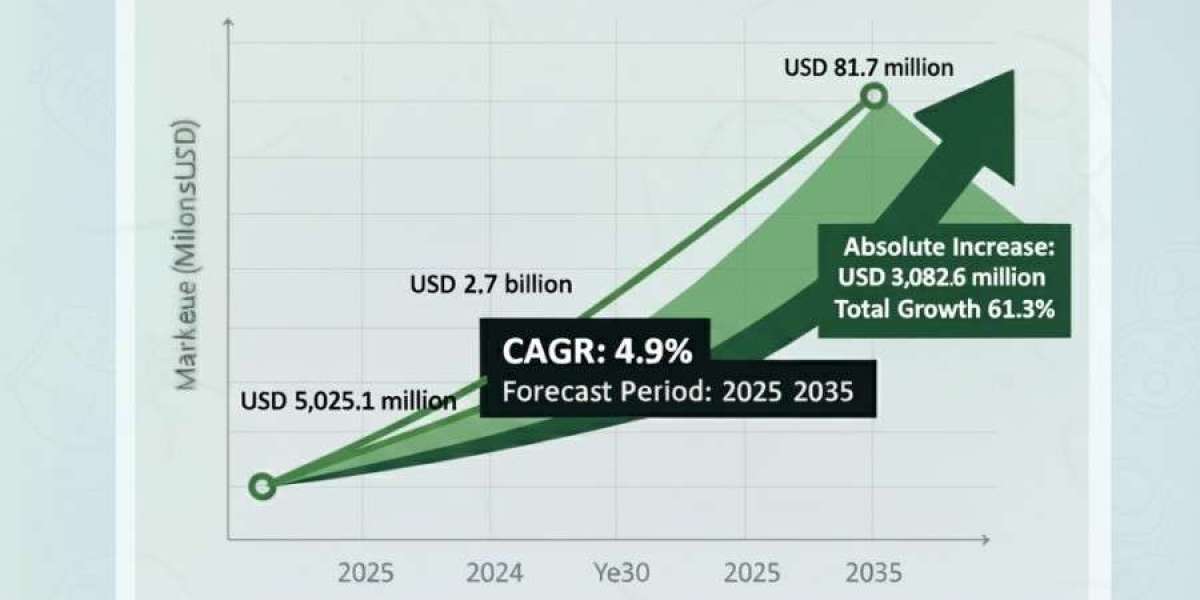
The question of whether Purerawz products are third-party tested is central to evaluating research compound integrity, batch consistency, and analytical transparency. In a market where product purity directly impacts experimental outcomes, the presence or absence of independent verification defines credibility.
This comprehensive analysis examines how Purerawz approaches third-party testing, what documentation to look for, how AICAR is verified for purity and potency, and how serious researchers should interpret Certificates of Analysis (COAs).
Understanding Third-Party Testing in the Research Compound Industry
Third-party testing refers to analytical verification conducted by an independent laboratory with no financial connection to the manufacturer or retailer. In the context of research compounds such as AICAR, this testing typically includes:
- High-Performance Liquid Chromatography (HPLC)
- Liquid Chromatography–Mass Spectrometry (LC-MS)
- Nuclear Magnetic Resonance (NMR)
- Fourier-Transform Infrared Spectroscopy (FTIR)
- Microbial and heavy metal screening
For a company like Purerawz, third-party testing means each batch of compounds is analyzed externally to confirm:
- Identity (correct molecular structure)
- Purity percentage
- Absence of contaminants
- Batch-specific consistency
Purerawz Quality Assurance Framework
Purerawz positions itself as a supplier focused on research-grade compounds. Its quality control process generally involves:
- Raw material sourcing verification
- In-house analytical screening
- External third-party laboratory testing
- Batch-level documentation
- Certificate of Analysis release
The presence of a batch-specific COA is the most critical indicator of third-party validation. A legitimate COA should include:
- Laboratory name
- Testing methodology
- Batch number
- Date of analysis
- Purity percentage
- Signature or verification stamp
Without these elements, claims of third-party testing lack substance.
AICAR Testing Standards: What High-Purity Verification Requires
AICAR (5-Aminoimidazole-4-carboxamide ribonucleotide) is a metabolic modulator widely used in research settings. Because AICAR influences AMPK pathways, purity deviations can significantly alter experimental data.
Essential Testing Criteria for AICAR
For AICAR supplied by Purerawz or any vendor, verification should confirm:
- ≥98% purity via HPLC
- Molecular mass confirmation via LC-MS
- Structural integrity via NMR
- Absence of residual solvents
- Heavy metal screening compliance
Batch Consistency
Consistency across batches is critical. A reputable supplier provides batch numbers that match:
- Product label
- COA documentation
- Internal tracking system
If the COA does not correspond to the exact batch purchased, third-party verification cannot be considered valid.
Analytical Methods Commonly Used in Purerawz Testing
1. High-Performance Liquid Chromatography (HPLC)
HPLC separates chemical components and determines purity percentage. For AICAR, a sharp single peak at the expected retention time confirms compound integrity.
2. LC-MS (Liquid Chromatography–Mass Spectrometry)
LC-MS verifies molecular weight and identifies trace impurities. This is essential for confirming AICAR’s molecular formula.
3. NMR Spectroscopy
NMR validates structural configuration. For compounds like AICAR, correct proton shifts confirm authenticity.
Transparency Indicators That Strengthen Purerawz Credibility
When evaluating whether Purerawz products are genuinely third-party tested, look for:
- Publicly accessible COAs
- Batch-specific documentation
- Independent laboratory branding
- Clear analytical methodology
- Recent testing dates (not recycled reports)
Companies that publish outdated or generic COAs undermine trust.
Why Third-Party Testing Matters for Research Accuracy
Inaccurate compound purity skews experimental outcomes. For metabolic research involving AICAR:
- Even 2–3% impurity can affect AMPK activation studies.
- Contaminants may introduce unaccounted biological activity.
- Reproducibility suffers when batch integrity is inconsistent.
Third-party testing eliminates these risks by validating compound composition through independent verification.
Comparing Self-Tested vs. Independently Tested Products
Criteria | Self-Tested Only | Third-Party Tested |
Bias Risk | High | Low |
Independent Validation | No | Yes |
Legal Accountability | Limited | Stronger |
Research Reliability | Variable | Consistent |
Documentation Transparency | Often Minimal | Detailed |
Third-party validation is the defining benchmark separating premium research suppliers from low-tier vendors.
Common Red Flags When Evaluating Purerawz or Any Supplier
- COAs without lab identification
- Missing batch numbers
- Identical reports reused across batches
- Purity claims without methodology
- Outdated documentation
Authentic third-party testing always includes traceability.
Best Practices for Researchers Purchasing AICAR from Purerawz
- Request the latest batch-specific COA before purchase.
- Confirm testing date aligns with production timeline.
- Verify analytical methods are listed.
- Ensure purity meets ≥98% threshold.
- Cross-check molecular weight via LC-MS confirmation.
These steps protect research integrity.
The Bottom Line on Purerawz Third-Party Testing and AICAR
Purerawz emphasizes third-party testing as part of its quality control process. However, verification must be performed at the batch level using authenticated Certificates of Analysis. For compounds like AICAR, independent laboratory validation through HPLC, LC-MS, and NMR is essential to ensure purity, potency, and reproducibility.
In the research compound industry, claims alone are insufficient. Documentation, transparency, and verifiable lab reports determine credibility. When these elements are present and batch-matched, third-party testing supports confidence in Purerawz product integrity.